About The Prosser Lab
The Prosser Lab – developing new therapies for the heart and brain.
(Simple, right?)
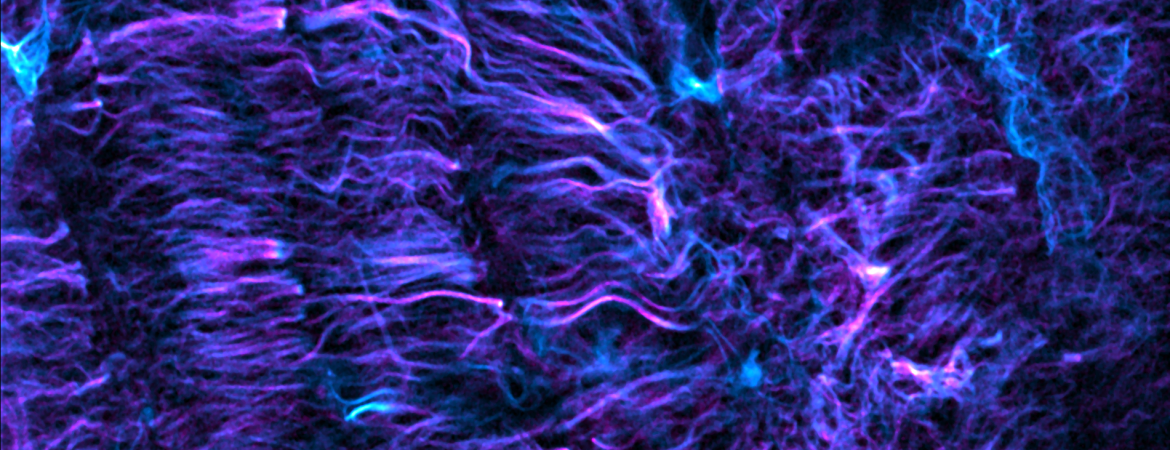
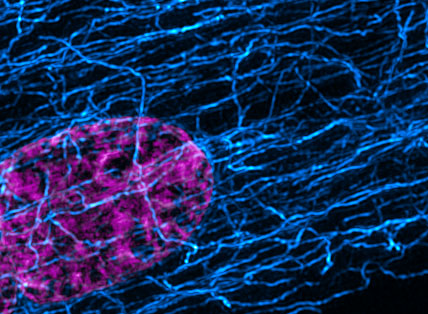
Our lab interrogates the forces of the heart – the mechanisms that control how a heart cell generates force, and how force itself feeds back to regulate cell function and form. We leverage cellular biophysics and engineering, super-resolution imaging and cardiology to tackle the fundamental challenges of the mechanobiology of the heart. Our goal is to provide transformative insight into how the heart beats, and to inform new therapies for the treatment of heart disease.
The diagnosis of Dr. Prosser’s daughter with STXBP1 encephalopathy also spurred the generation of a new therapeutic arm in the lab for neurological applications. We are developing novel, anti-sense oligonucleotide based therapies for the treatment of inherited neurodevelopmental disorders, such as those caused by mutations in STXBP1.
The Prosser Lab – developing new therapies for the heart and brain.
(Simple, right?)
Our lab interrogates the forces of the heart – the mechanisms that control how a heart cell generates force, and how force itself feeds back to regulate cell function and form. We leverage cellular biophysics and engineering, super-resolution imaging and cardiology to tackle the fundamental challenges of the mechanobiology of the heart. Our goal is to provide transformative insight into how the heart beats, and to inform new therapies for the treatment of heart disease.
The diagnosis of Dr. Prosser’s daughter with STXBP1 encephalopathy also spurred the generation of a new therapeutic arm in the lab for neurological applications. We are developing novel, anti-sense oligonucleotide based therapies for the treatment of inherited neurodevelopmental disorders, such as those caused by mutations in STXBP1.